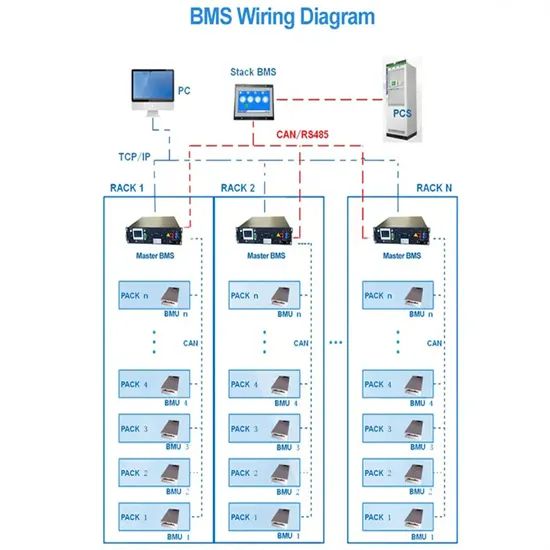
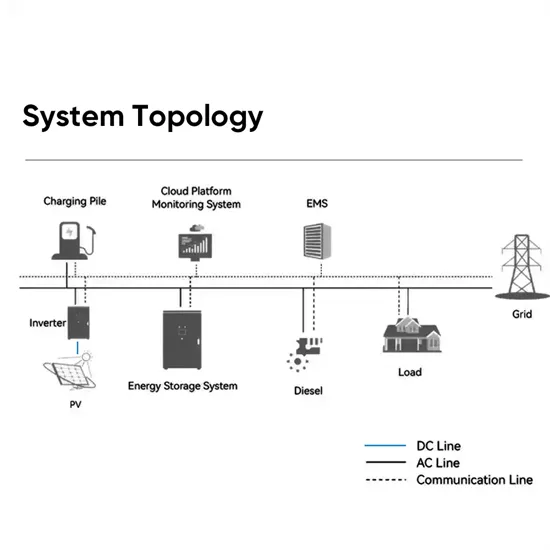
Thermodynamic Analysis of Lithium''s Ionotropism at Low
Explanation Melting Point Example: Lithium has a melting point of approximately 180.5°C. This calculator checks whether an input temperature is above or below this value and determines if Lithium is in a